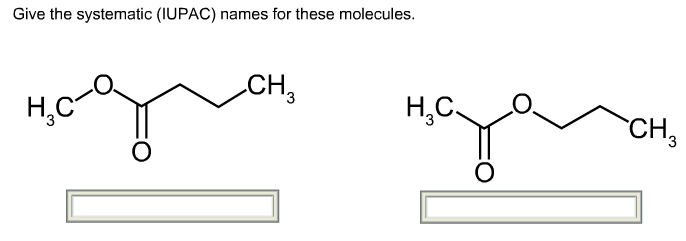
Examples: Fe 2+ and Fe 3+, Pb 2+ and Pb 4+.įor type II compounds the charge of the metal ion must be specified, and this is systematically done with Roman numeral indication.Įxample: iron(II) for Fe 2+ and iron(III) for Fe 3+. Type II binary ionic compounds can form more than one type of cation. 
Examples of naming of compounds type I is given in the table: Type I compounds, naming examples Type I binary ionic compounds contains a metal that forms only one type of cation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
